Depo-Provera® Birth Control Shot Lawsuit
Brain Meningioma After Long-Term Use Of Depo shots? Learn About Your Legal Options
If you or a loved one are seeking information about a Depo lawsuit or Depo shot lawsuit, you may be eligible to request a confidential case review related to alleged health risks associated with long-term Depo-Provera® use.
Start My Free Case Review The information on this page is not medical advice. Any references to Depo‑Provera and health conditions are based on allegations made in legal claims and litigation. No medical conclusions or diagnoses are implied.Lawsuits involving the Depo brain tumor lawsuit allege that prolonged use of Depo-Provera® (injectable medroxyprogesterone acetate) may be associated with an increased risk of certain meningiomas, and that patients may not have been adequately warned.
These claims are part of broader birth control lawsuit filings currently under review in federal court.

You Relied on a Birth Control Shot. Now You’re Facing a Serious Diagnosis.
According to published research, including a large observational study reported in The BMJ, prolonged exposure to injectable medroxyprogesterone acetate has been associated with a higher observed risk of intracranial meningioma.
Based on these findings, individuals have filed Depo-Provera lawsuits and Depo class action lawsuit claims alleging inadequate warnings regarding long-term risks.
If you:
- Used brand Depo-Provera® for at least one year (4+ injections), and
- Were later diagnosed with a brain or head meningioma,
you may meet the Depo shot lawsuit qualifications for an attorney review.
Federal cases have been centralized for coordinated pretrial proceedings. Defendants deny liability, and litigation is ongoing.
Start My Free Case ReviewYour Privacy Comes First

100% Confidential Intake

Secure submission of information

Attorney review of your medical history
Some attorneys may offer representation on a contingency basis. Fee arrangements depend on the attorney and the facts of the case.
Request a Case Review in About 60 Seconds
To help screen for eligibility and avoid misinformation, our intake uses open-ended questions rather than simple yes/no answers. This helps confirm whether your medical history aligns with current Depo lawsuit update criteria.
Do You Have a Claim?
You may be eligible for a Depo class action or individual Depo shot lawsuit review if all of the following apply:
(Arachnoid tumor may qualify; arachnoid cyst typically does not.)
Request a Case ReviewMeeting the described criteria does not guarantee eligibility, legal representation, or compensation. All cases are reviewed individually by licensed attorneys.
Our 3-Step Process
Secure & Respectful Intake
Share your medication history and diagnosis details through a confidential form.
Medical & Records Review
Your information may be reviewed for timeline, diagnosis type, and eligibility criteria.
Legal Review & Next Steps
If appropriate, a licensed attorney may contact you to discuss potential legal options related to the Depo lawsuit.
No outcome is guaranteed. Results depend on individual facts and applicable law.
What These Lawsuits Allege About Depo-Provera®
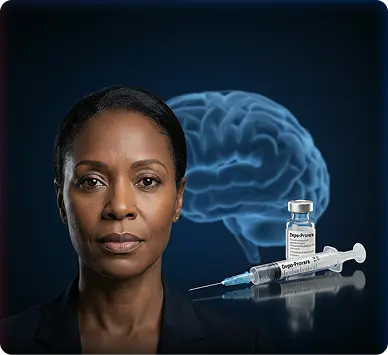
Current Depo shot lawsuit filings generally focus on whether manufacturers adequately warned patients and healthcare providers about potential meningioma risks associated with long-term use.
The litigation is active and evolving. Defendants deny wrongdoing. Regulatory and labeling discussions, including reported FDA-related updates, are part of ongoing review.
What Legal Claims May Address
A meningioma diagnosis can involve significant medical care and lifestyle changes. Depending on individual circumstances, some claims may seek to address:
- Medical expenses
- Lost income
- Other documented impacts
Submitting information allows an attorney to explain whether legal options may be available.
(No outcome is guaranteed. Results depend on individual facts.)
Understanding Depo Shot Side Effects and Symptoms
Meningiomas are often described as slow-growing, but symptoms can still be serious depending on size and location. Reported symptoms may include:
- Seizures
- Vision or hearing changes
- Chronic headaches
- Neurological deficits
Treatment may involve monitoring, surgery, or radiation based on medical advice.
Take the Next Step:
If you believe your history may meet Depo shot lawsuit qualifications, you can request a free review today.
- No upfront fees
- Confidential process
- No obligation to proceed
Why Women Choose This Review

Completely Confidential

No Upfront
Costs

Fast Eligibility Screening
 Act Promptly: Time Limits May Apply
Act Promptly: Time Limits May Apply
Even when national litigation is active, each person’s claim can have deadlines. It’s best to confirm eligibility sooner rather than later
Start Eligibility CheckFAQs
I used the shot years ago, can I still qualify?
Possibly. The key is at least 1 year of brand Depo-Provera® use and a qualifying meningioma diagnosis after that minimum use.
